- Lab Consulting
Set up, support and training for your lab
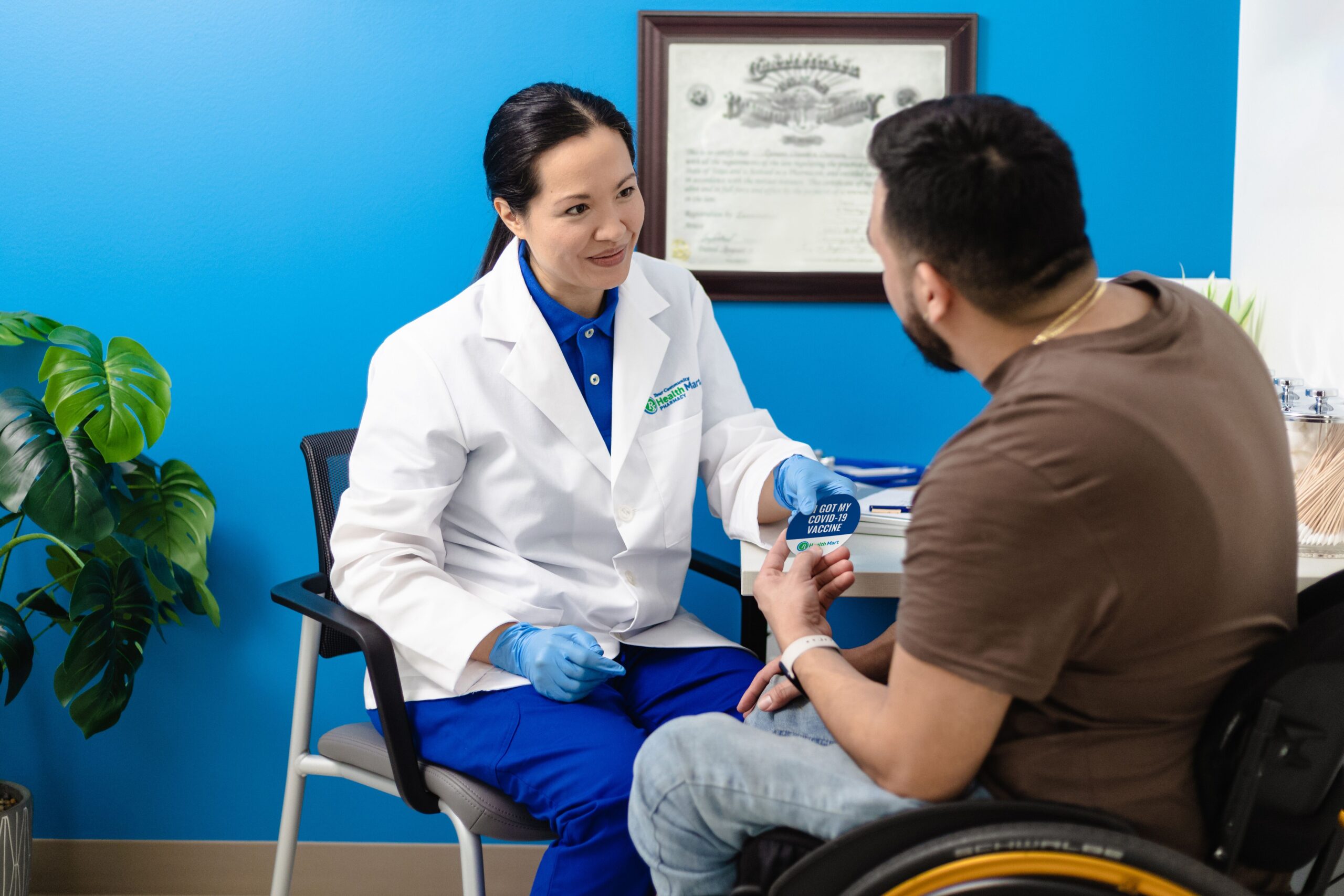
Improve clinical efficiency with help from licensed professionals

Process and Compliance Assistance
Whether you are starting a new laboratory or have an existing laboratory, you can benefit from a knowledgeable consultant. A consultant, whose familiarity with the complexities and licensing processes, can assist in your lab’s compliance with CLIA, COLA, CAP, IQCP, PPM, state and federal regulatory agencies.

In-depth guidance
We understand that lab is a long-term business strategy that requires a seasoned partner. From setup to ongoing laboratory consulting and compliance training, our laboratory consultants are credentialed, licensed and have the specialized clinical laboratory expertise to support your laboratory testing needs regardless of size, capacity or complexity.
How MedSol Can Help
Ensure proper license and regulatory documentation
Technical expertise and regulatory management
Support with enrollment and maintenance of quality assurance
Provide audit and onsite support to ensure standards
Technical support and consultation
Evaluate and assess personnel for compliance certification
Support laboratory builds, instrument set-ups and validations
Lab setup
MedSol’s lab setup option provides assistance with licensure, personnel verification, development of policy and procedure, QA plans, validation study development and general guidance throughout lab setup.
The reference lab setup option includes a customized CD for client services directory according to CLIA requirements. MedSol’s lab consulting includes ongoing support with monthly calls or site visits to discuss and assess quality control, maintenance, proficiency and quality assessment for continued compliance in the laboratory setting.

Lab Setup Options Include:
- CLIA licensure or upgrade applications
- State-specific licensure application
- Enrollment in proficiency testing
- Evaluation of laboratory director and personnel qualifications
- Enrollment in laboratory director course
- Enrollment with accreditation organization if desired
- Compilation of general laboratory policy and procedure manual
- Compilation of assay-specific policy
- Compilation of a complete quality assessment plan
- General laboratory safety policy
- Data reduction with validation of analyzers
- All logs for reagent inventory, temperature, QC and patient testing
- General guidance for proper CPT coding
- Preparation for CLIA or accreditation organization survey and inspection process
Featured Product Categories
Lab consulting and technical support
Whether on-site or by phone, fax or email, MedSol’s ongoing lab consulting provides you with a choice of monthly, bi-monthly, quarterly or six-month review of all quality control, quality assessment, calibration, maintenance, proficiency testing and personnel records. Ongoing consulting updates you on CLIA and state regulatory changes. MedSol’s licensed professionals can help ensure your lab is efficient, compliant and continues to provide quality testing.

Ongoing Consulting Options:
- Monthly review of quality control
- Monthly review of quality assessment plan
- Schedule for performance of calibration and calibration verification
- Data reduction for six-month calibration verification studies
- Review of proficiency testing with remedial action as warranted
- Notification for renewal of licensure
- Updates for all CLIA and state-specific regulatory requirements
- Assistance with validation studies for new testing
- Patient population reference range verification
- Update to policy and procedure manual as warranted
- Annual competency reviews
Laboratory strategy and management
MedSol support includes many support services designed to help you provide continued quality testing. These services ensure your lab is efficient and financially healthy with a commitment to assist in improving patient outcomes. Medsol consultants can provide:
- Introduction to lab training course
- On-site mock inspections
- Policy and procedure manuals
- Post inspection correction and restorative processes


Get the setup or ongoing support you need for your laboratory
Be advised that information contained herein is intended to serve as a useful reference for informational purposes only and is not complete clinical information. This information is intended for use only by competent healthcare professionals exercising judgment in providing care. McKesson cannot be held responsible for the continued currency of or for any errors or omissions in the information.
The product information contained in this document, including the product images and additional product materials, was collected from various supplier sources. All product claims and specifications are those of the product suppliers and have not been independently verified by McKesson Medical-Surgical or its affiliates (“McKesson”). McKesson is not responsible for errors or omissions in the product information. The properties of a product may change or be inaccurate following the posting or printing of the product information in the document, either in the print or online version. Caution should be exercised when using or purchasing any products from McKesson’s online or print documents by closely examining the product packaging and the labeling prior to use. Due to product changes, information listed in this document is subject to change without notice. This information is placed solely for your convenience in ordering and McKesson disclaims all responsibility for its completeness and accuracy, whether or not the inaccuracy or incompleteness is due to fault or error by McKesson.
©2024 McKesson Medical-Surgical Inc. All trademarks and registered trademarks are the property of their respective owners.